Abstract
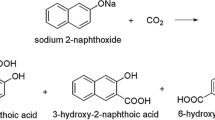
The mechanisms of the carboxylation reactions of lithium and sodium phenoxides are investigated by means of the DFT method with a CEP-31 + G(d) basis set. The introduction of diffusion functions does not affect the outcome of the calculations. As a consequence, the results of this investigation are in good agreement with the findings obtained by means of the LANL2DZ basis set. Lithium phenoxide yields only salicylic acid in the Kolbe-Schmitt reaction. The reaction of sodium phenoxide can proceed in the ortho and para positions, but the para-substituted product can be expected at a very low concentration in the reaction mixture. The deviation of lithium and sodium phenoxides from the mechanisms of carboxylations of other alkali metals is a consequence of the small ionic radii of lithium and sodium.
Similar content being viewed by others
References
H. Kolbe, Liebigs Ann. 113, 125 (1860).
H. Kolbe, and E. Lautemann, Liebigs Ann. 115, 157 (1860).
R. Schmitt, J. Prakt. Chem. 31, 397 (1985).
M. Windholz, The Merck Index, 10th ed. (Merck Inc., Rahway, 1983), p. 8190.
J. E. F. Reynolds, in Martindale the Extra Pharmacopoeia, 31st ed. (The Royal Pharmaceutical Society, London, 1996), pp. 1093–1105.
A. S. Lindsey and H. Jeskey, Chem. Rev., p. 583 (1957).
M. Kunert, E. Dinjus, M. Nauck, and J. Sieler, Chem. Ber./Recueil 130, 1461 (1997).
Y. Kosugi, Y. Imaoka, F. Gotoh, et al., Org. Biomol. Chem. 1, 817 (2003).
Z. Marković, J.P. Engelbrecht, and S. Marković, Z. Naturforsch, A: Phys. Phys. Chem. Kosmophys. 57 812 (2002).
I. Stanescu and L. E. K. Achenie, Chem. Eng. Sci. 61, 6199 (2006).
I. Stanescu, R. R. Gupta, and L. E. K. Achenie, Mol. Simul. 32, 279 (2006).
Z. Marković, S. Marković, and N. Begović, J. Chem. Inf. Model. 46(5), 1957 (2006).
Z. Marković, S. Marković, and N. Begović, in 8th International Conference on Fundamental and Applied Aspects of Physical Chemistry (Belgrade, 2006), C-6-P.
A. D. Becke, Phys. Rev. A: At., Mol., Opt. Phys., p. 2098 (1988).
C. Lee, W. Yang, and R.G. Parr, Phys. Rev. B: Condens. Matter, p. 785 (1988).
A. D. Becke, J. Chem. Phys. 98, 5648 (1993).
M. J. Frisch, G. W. Trucks, H. B. Schlegel, et al., Gaussian 98, Revision A.9 (Gaussian Inc., Pittsburg, PA, 1998).
Chemical Applications of Density Functional Chemistry, Ed. by A. Laird, R. B. Ross, and T. Zeigler (American Chemical Society, Washington, DC, 1996).
W. J. Stivens, H. Bach, and J. Krauss, J. Chem. Phys. 81, 6026 (1984).
J. P. Foster, and F. Weinhold, J. Am. Chem. Soc. 102, 7211 (1980).
A. E. Reed, R. B. Weinstock, and F Weinhold, J. Chem. Phys. 83, 735 (1985).
G. Schaftenaar, MOLDEN 3.7 (CAOS/CAMM Center, The Netherlands, 1998).
W. J. Hehre, A. J. Shusterman, and W. W. Huang, A Laboratory Book of Computational Organic Chemistry (Wavefunction Inc., Irvine, CA, 1996).
J. A. Marti, Chem. Phys. 265, 263 (2001).
O. Baine, G. F. Adamson, J. W. Barton, et al., J. Org. Chem. 19, 510 (1954).
K. Suenobu, M. Nagaoka, and T. Yamabe, THEOCHEM 461–462, 581 (1999).
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Marković, S., Marković, Z., Begović, N. et al. Mechanism of the Kolbe-Schmitt reaction with lithium and sodium phenoxides. Russ. J. Phys. Chem. 81, 1392–1397 (2007). https://doi.org/10.1134/S0036024407090087
Issue Date:
DOI: https://doi.org/10.1134/S0036024407090087